Quarterly Results Report

In Q1 the Council for Responsible Nutrition team focused on issues that matter most to dietary supplement and functional food member companies: protecting the ability to bring products to market, calling for regulatory clarity, and promoting scientific credibility. CRN’s work this quarter reflects a clear priority—ensuring the industry can operate within a stable, science-based framework while continuing to innovate.
Download printable PDF summary of Q1 2026 report.
Q1 2026 AT A GLANCE — CRN:
- Petitioned U.S. Supreme Court to review potentially precedent-setting free speech case
- Influenced FDA thinking on next-generation ingredients
- Opposed state legislation to prevent fragmented regulation
- Delivered real-time tariff and trade guidance
- Published symposium report underscoring growing scientific consensus that nutrition should be recognized as a foundational component of healthcare
- Reinforced consumer trust through science and media engagement
- And more—read on for details and links
PROTECTING MARKET ACCESS AND REDUCING BUSINESS RISK
CRN petitions U.S. Supreme Court to review New York law restricting supplement sales based on marketing claims
In March, CRN petitioned the U.S. Supreme Court to review a New York law restricting the sale of certain dietary supplements to minors based solely on how those products are labeled or marketed. This New York law is a content-based infringement on speech and is wholly different than laws that limit the sales of products based on their ingredients or demonstrated harm. The law broadly sweeps into its prohibition products that are safe, beneficial, and have never been shown to harm minors.
CRN’s petition urged the Court to reaffirm First Amendment protections for commercial speech and to resolve a growing split among federal appellate courts regarding the evidentiary and judicial standards required to justify government restrictions on truthful, non-misleading communications.
“This case goes to the heart of how the government can restrict commercial speech and the high bar that must be upheld for content-based speech restrictions,” explained SVP and General Counsel Megan Olsen.
A decision could set precedent for how supplements are marketed and sold across all 50 states—important with the proliferation of similar bills across the U.S. Read more in CRN's press release and CRN Supplement newsletter coverage.
CRN promotes responsible innovation at FDA public meeting

At a pivotal moment for the future of dietary supplement innovation, two voices from CRN helped bring clarity—and urgency—to the conversation at FDA's March 27 public meeting on the evolving scope of dietary ingredients (Docket FDA-2026-N-2047).
CRN SVP and Chief Science Officer Andrea Wong, Ph.D., and SVP and General Counsel Megan Olsen delivered testimony that, together, bridged science, law, and practical reality in a way that reflects the unique value CRN provides.
Dr. Wong grounded the discussion in what innovation actually looks like today. From precision fermentation to next-generation probiotics, the science has moved far beyond 1994—but regulatory interpretation hasn’t always kept pace. Her message was clear: we cannot define “dietary substance” so narrowly that it excludes the very innovations DSHEA was designed to accommodate. As she put it, responsible industry isn’t looking for shortcuts—it’s “knocking at the front door,” ready to demonstrate safety through the appropriate pathways, not forced into inefficient or impractical workarounds.
Ms. Olsen then reinforced that position with legal precision. The statute’s language—“to supplement the diet by increasing the total dietary intake”—was intentionally expansive. It does not require prior presence in the food supply, nor does it limit innovation to what existed decades ago. Legislative history confirms this was a conscious choice by Congress to allow the category to evolve alongside science. The path forward, as she emphasized, is not to rewrite the law—but to interpret it as written, in a way that supports both innovation and safety.
Together, their testimony reflects what CRN has consistently stood for:
✔️ Science-based policy
✔️ Regulatory clarity
✔️ Responsible innovation
✔️ A viable pathway (NDI) that actually works
As Dr. Wong noted, the detailed questions FDA is asking about identity, manufacturing, and composition are absolutely valid—but they belong in the evaluation of safety, not as a gatekeeping mechanism for whether an ingredient qualifies in the first place.
This distinction matters because when the framework is too narrow, innovation doesn’t stop—it just moves elsewhere, often outside the very oversight system designed to protect consumers. A more inclusive, science-aligned interpretation does the opposite: it brings innovation into the regulatory system, where it can be properly evaluated. That’s the opportunity in front of FDA now. And it’s why this moment feels different.
Read more about the FDA public meeting in the CRN Supplement newsletter.
CRN provides tariff and trade guidance, resources
CRN kept members updated on tariff news related to the International Emergency Economic Powers Act (IEEPA), including a Court of International Trade (CIT) amendment with good news for industry on refund procedures after the Supreme Court invalidated the President's tariff authority. The amendment clarified that U.S. Customs and Border Protection (CBP) must reliquidate and refund IEEPA tariffs even for entries beyond for which liquidation is final. These are entries that are no longer within the normal protest or voluntary reliquidation window. CBP expressed its refund functionality remained on track for mid-April and confirmed it plans to roll out refunds in phases, including a later phase for entries that have already reached final liquidation.
During Q1, CRN provided critical guidance on evolving tariff policies, including a members-only briefing with trade expert Brooke Ringel of Kelley Drye, on retainer to support CRN member queries, and an industry-wide webinar on 2026 tariff implications, with Ringel and other experts, available on demand for members here.
CRN met with representatives from the U.S. Department of Commerce in March to advocate for the exclusion of dietary supplement and food ingredients from any tariff actions under the Section 232 pharmaceutical investigation. Read more in the CRN Supplement member newsletter.
ADVANCING POLICY THAT WORKS
CRN drives smart, consistent policy to prevent a patchwork of state laws and protect a viable national marketplace
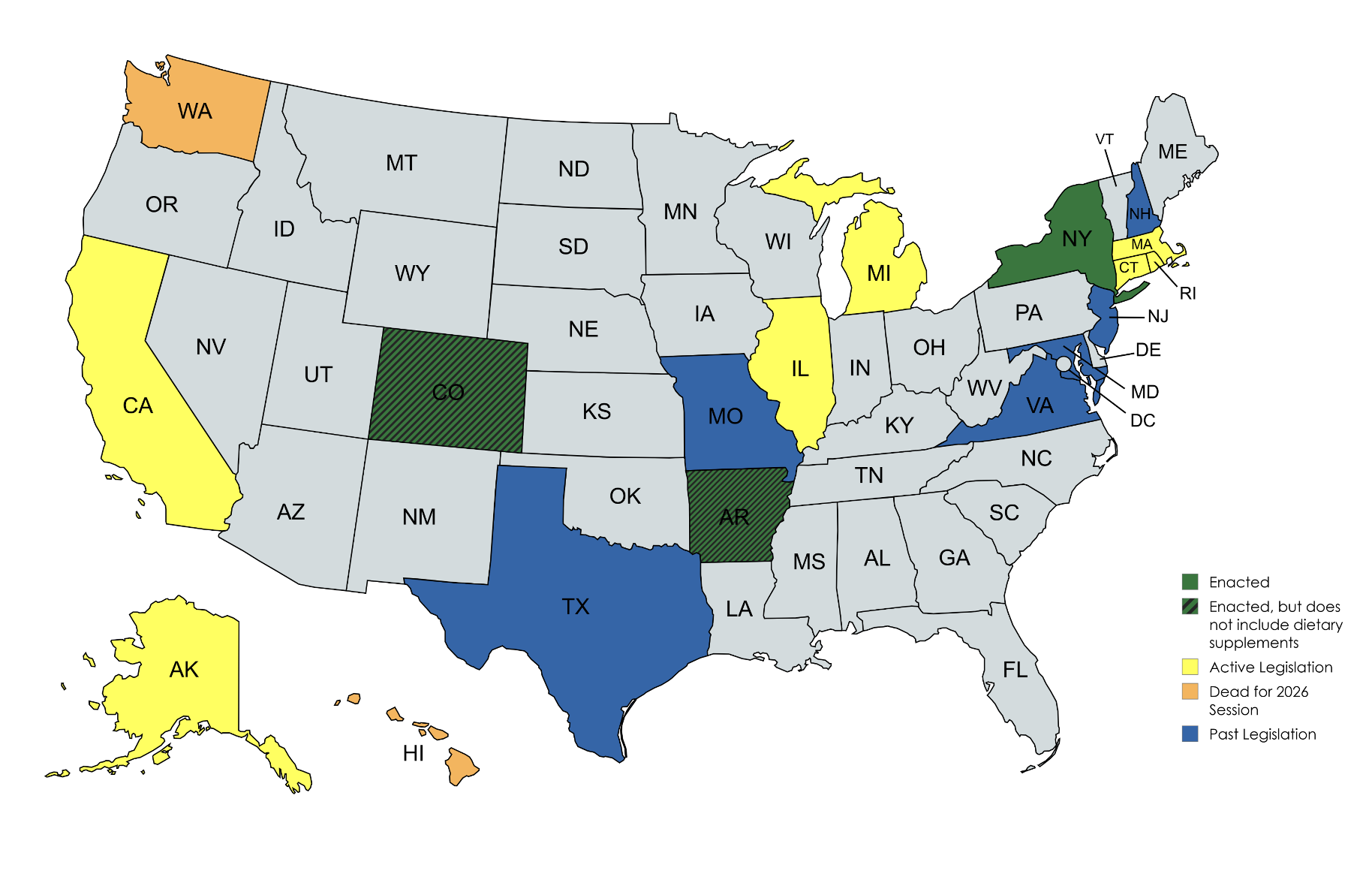 During Q1, CRN engaged on the growing wave of state legislative activity, including opposing California’s protein powder bill and Virginia’s supplement disclosure requirements creating inconsistent or misleading standards, instead advocating for a national, science-based approach.
During Q1, CRN engaged on the growing wave of state legislative activity, including opposing California’s protein powder bill and Virginia’s supplement disclosure requirements creating inconsistent or misleading standards, instead advocating for a national, science-based approach.
At the federal level, CRN expressed its strong support of the Dietary Supplement Regulatory Uniformity Act, legislation that reaffirms the FDA’s authority as the national regulator of dietary supplements and pre-empting conflicting state laws.
CRN welcomed introduction of the Dietary Supplement Listing Act of 2026, marking a significant milestone in CRN’s years-long effort to modernize oversight of the dietary supplement marketplace through a federal registry.
CRN also monitored and activated on emerging GRAS reform proposals, ingredient safety scrutiny, and overall FDA priority-setting.
In addition, CRN joined the kickoff call of the Recycling Leadership Council as the only participating dietary supplement trade association. RLC is a cross-industry coalition focused on establishing clear, consistent national recycling definitions to reduce plastic waste.
Convening the community and keeping members up to date
CRN presented a Member Breakfast Briefing at Expo West, sponsored by Nuritas, RT Specialty, Kaneka Nutrients, and Pharmavite/Nature Made, with Senior Director, Government Relations Michael Meirovitz providing an update on the evolving federal and state political landscape impacting the dietary supplement industry. (CRN members, access his presentation deck here.) Meirovitz highlighted several key federal priorities, including efforts to allow consumers to use HSA, FSA, and HRA funds to purchase dietary supplements, as well as progress on dietary supplement product listing legislation aimed at improving transparency and strengthening FDA oversight of the marketplace. State-level legislation in play was also covered, including proposals related to age restrictions, ingredient bans, labeling and testing disclosures, and potential changes to GRAS policies.
ELEVATING INDUSTRY LEADERSHIP
Through scientific and trade press publications, bylines, podcasts, and expert engagement, CRN amplified the industry’s voice on:
- Drug preclusion reform
- Product listing as a pro-consumer solution
- Clarifying supplement misinformation
- Scientific consensus supporting nutrition as fundamental to wellness
Mister talks MAHA impact on supplement market at ECRM symposium
CRN President & CEO Steve Mister spoke at the ECRM GLP1 Total Wellness Symposium, discussing what the Make America Healthy Again (MAHA) movement means for the dietary supplement market. (CRN members, access his presentation deck here.)
Why it matters: MAHA’s focus on chronic disease prevention and nutrition is creating opportunity for supplements, even as early policy signals remain mixed, but with persistence, patience, and pragmatism, CRN is advocating for change that protects responsible industry and best serves consumers.
Pressure points include:
- Ongoing scrutiny of self-GRAS pathways used for new ingredient development.
- Tariffs affecting globally sourced raw materials.
- A growing patchwork of state-level age restrictions and additive bans.
- Drug preclusion decisions that favor pharmaceutical interests over supplement innovation.
Mister encouraged industry stakeholders to lean into prevention and nutrition messaging by:
- Emphasizing the role of supplements in addressing documented nutrient gaps.
- Supporting the “Eat Real Food” message—while recognizing most Americans fall short nutritionally.
- Demonstrating high-quality manufacturing, safety, and substantiation
- Highlighting barriers that limit consumer access.
- Emphasizing research showing supplementation’s potential to reduce healthcare spending.
CRN’s policy focus: To ensure supplements are positioned as part of the solution, CRN continues advancing policy priorities that protect responsible industry and best serve consumers. These include:
- A federal supplement registry (Mandatory Product Listing) to improve market visibility.
- Stronger FDA enforcement against bad actors.
- Federal preemption to prevent inconsistent state laws.
- Resolution of drug preclusion concerns.
- Tariff relief to maintain affordability and access.
- Inclusion of supplements as eligible FSA/HSA expenses.
Bottom line: The objective isn’t more regulation—it’s smarter regulation. A predictable, science-based framework protects responsible companies, supports innovation, and strengthens consumer trust.
CRN attends National Conference on Women’s Health

CRN was invited to attend the 2026 National Conference on Women’s Health, held March 11–13 in Washington, D.C., convening leaders from government, academia, healthcare, and industry to discuss emerging issues shaping women’s health. Dr. Andrea Wong and Haiuyen Nguyen's participation provided an opportunity to stay informed on broader women’s health priorities and engage with stakeholders working across the healthcare ecosystem. Read more in the CRN Supplement member newsletter.
STRENGTHENING TRUST THROUGH SCIENCE
CRN reinforces evidence-based supplement safety
CRN released a review reaffirming that supplemental calcium is not associated with increased risk of cardiovascular disease, countering years of confusion fueled by flawed and misinterpreted studies. The review is part of updates to the CRN Vitamin and Mineral Safety publication, systematically evaluating the safety of all essential vitamins and minerals as well as other bioactive compounds by evaluating the latest clinical research.
CRN pushes back on misrepresentation of supplements in press coverage
CRN also responded to misleading media narratives—from the New York Times to Freakonomics and more, ensuring accurate, contextualized information reaches consumers. Be sure to follow us on LinkedIn to stay updated on our efforts to correct misinformation).
GROWING OUR MEMBERSHIP
In Q1, CRN welcomed new members Dream Big Ventures, IFF Health Sciences, Ingredients Online, Nutracom, and Vireo Systems, along with new associate members Circana, Certo, The Acheson Group (TAG), and The Ginger Network (TGN). Learn more in our press release.
Not yet a CRN member? Join our community and support the leading trade association of the dietary supplement and functional food industry.



